AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Bio rad western blot troubleshooting2/11/2024 
The time needed to view the stain on the blot also varies with the dye used. Other relatively expensive blot stains, such as SYPRO Ruby or amido black, are also used for blot quantitation and require special disposal methods. However, the ephemeral nature of the binding of the dye to the protein causes the intensity of the bands on the membrane to decrease rapidly, making detection and quantitation harder. Ponceau S staining is fast and relatively inexpensive, and the stained membrane can then be used for downstream applications such as western blotting. A common standard practice to verify protein transfer is staining the blot with Ponceau S, a negatively charged stain that binds to all positively charged amino acids in a protein. The proper transfer of proteins to the membrane is critical to the western blotting process. In contrast to Coomassie staining, Stain-Free technology is compatible with most downstream applications, such as western blotting, which helps not only when resources are limited, but also in the appropriate normalization of data for quantitation using total protein normalization (see Normalization of Data in Quantitative Western Blotting below). This is a serious drawback when the ultimate goal is the identification or quantitation of proteins in western blotting, especially when large gels and sample volumes are required for separation and visualization. The Coomassie dye-based method does not allow the same gel to be used for transfer during western blotting or for mass spectrometry. A. HeLa cell lysate dilutions from 80–2.5 µg total protein B. HeLa cell lysate dilutions from 20–1 µg total protein.Ĭompatibility with Downstream Applications Linear dynamic range provided by Stain-Free technology for total protein measurements. 
Stain-Free gels provide a linear dynamic range between 10 and 80 µg of total protein load from cell or tissue lysates at a higher range of protein load (Figure 2A) and from 20 to 1 µg at a lower range (Figure 2B) (Taylor et al. Ideally, the protein load should fall within the quantitative linear dynamic range of the antibody used for its detection (Taylor and Posch, 2014). The linear dynamic range for protein quantitation is defined as the range through which the signal intensity on a blot proportionally increases with the increase in protein load. Stain-Free gels have more reproducible data with smaller coefficients of variation compared to Coomassie or silver stains (McDonald et al. Some fluorescent stains can detect proteins at levels below the 1 or 0.5 ng limit. The limit of detection for the Stain-Free gels is 8 to 28 ng, similar to that of silver stains (0.6 to 1.2 ng), while Coomassie R-250 stain can detect protein amounts of at least 35 to 50 ng. 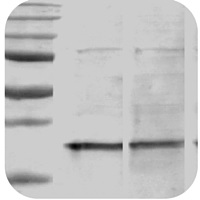 
The gel was imaged with a Stain-Free enabled imager, then stained with Coomassie (CBB R-250 and Bio-Safe G-250) stain and imaged on a densitometer. Serial 1:2 dilutions of broad range unstained molecular weight standards were separated on a 4–20% Criterion Stain Free Tris-HCI Gel. Comparison of a Stain-Free gel, CBB R-250, and Bio-Safe G-250 stained gel images. For proteins with higher tryptophan content, Stain-Free gels provide much higher sensitivity than CBB-stained gels (Figure 1).įigure 1. In general, the sensitivity of Stain-Free gels when visualizing data is equal to that of Coomassie-stained gels for all proteins. Protein visualization data obtained from Stain-Free gels are comparable to those obtained from gels stained with other dyes. Stain-Free Technology Provides More Sensitivity and Better Dynamic Range than Coomassie Stains Stain-Free imaging allows for the elimination of the inherently problematic use of housekeeping proteins as loading controls on western blots, enabling the user to obtain truly quantitative western blot data by normalizing bands to total protein in each lane.Īdvantages of Using Stain-Free Technology With the fluorophore covalently bound to the protein molecules, they can be imaged repeatedly on a gel or membrane after transfer, without additional staining and destaining steps. The addition of the fluorophore allows visualization of proteins in the gel and following transfer onto a membrane during western blotting but does not interfere with electrophoresis or downstream steps. This trihalo compound is covalently bound to tryptophan residues, enhancing their fluorescence when exposed to UV light, enabling the detection of proteins at levels as low as 10–25 ng. Stain-Free imaging technology utilizes a polyacrylamide gel containing a proprietary trihalo compound to make proteins fluorescent directly in the gel with a short photoactivation, allowing the immediate visualization of proteins at any point during electrophoresis and western blotting.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
